Valence Shell Electron Pair Repulsion Theory (VSEPR)
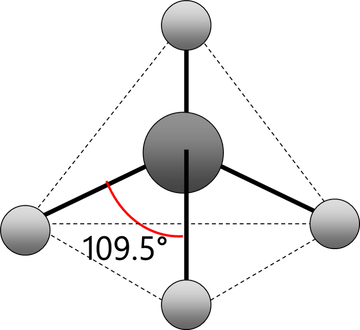
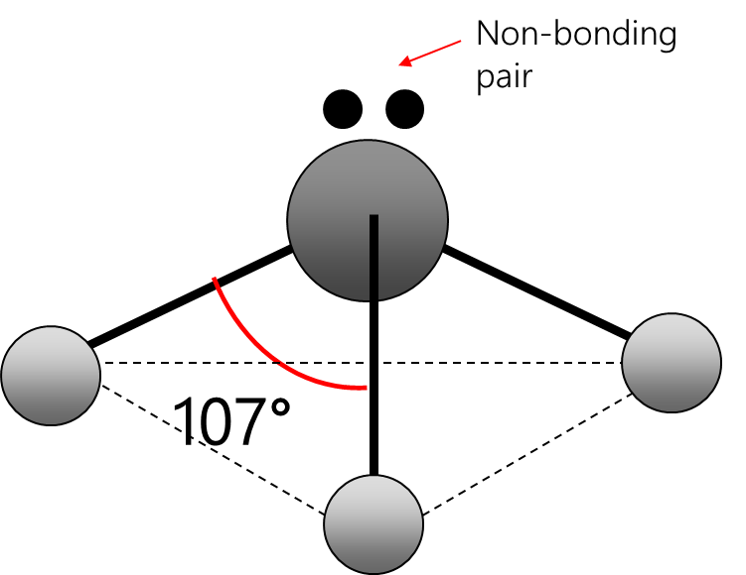
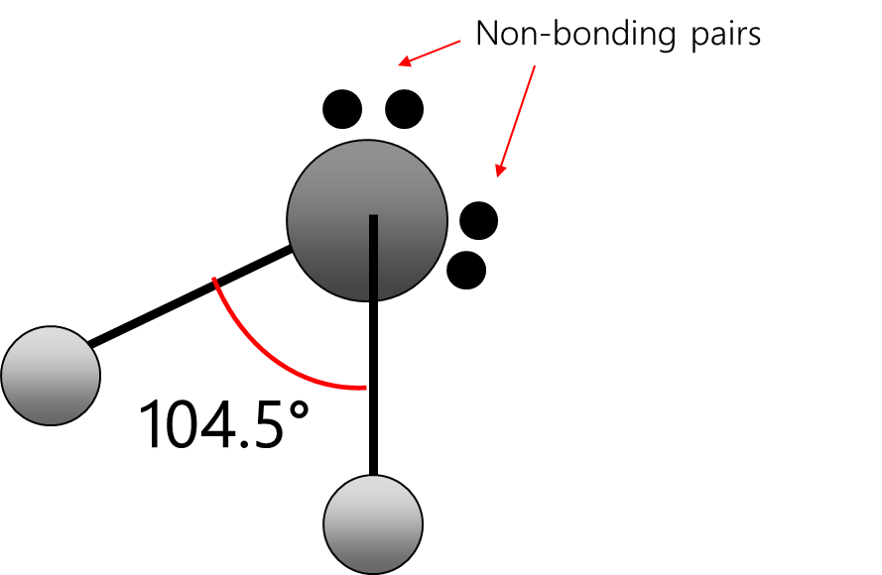
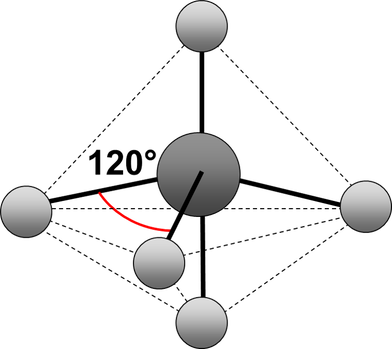
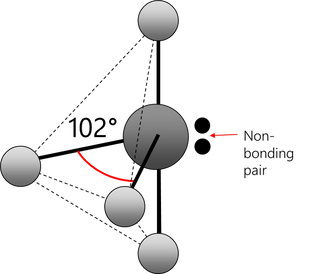
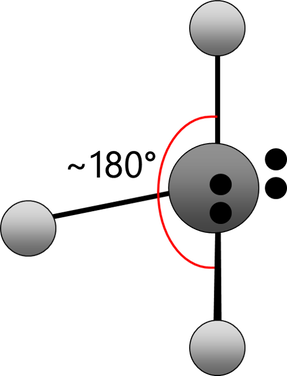
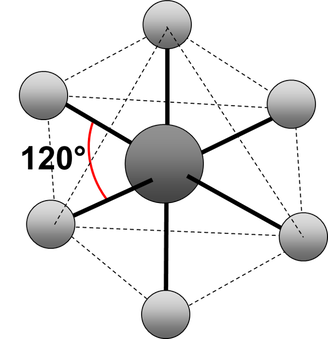
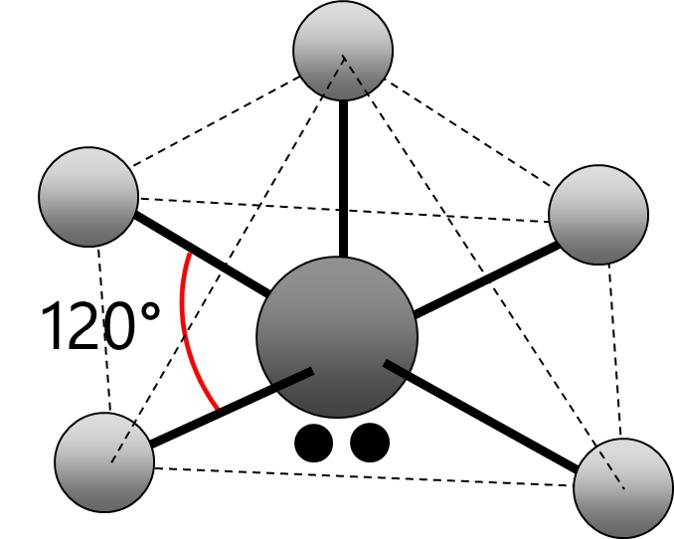
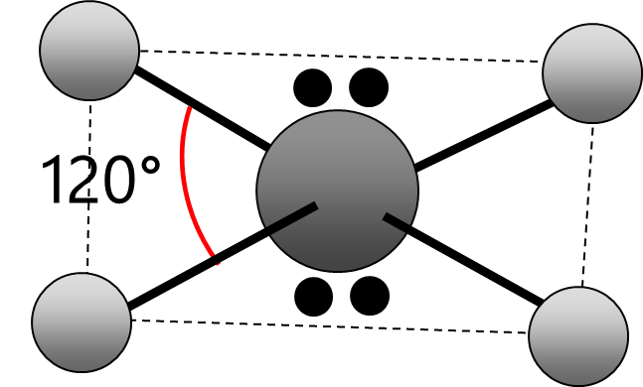
Electrons repel and therefore regions of negative charge, consisting of a pair of electrons, either bonded or non-bonded, move as far apart from each other as possible around a central atom. This enables electron repulsion to be minimised. This theory is used to predict molecular shape.
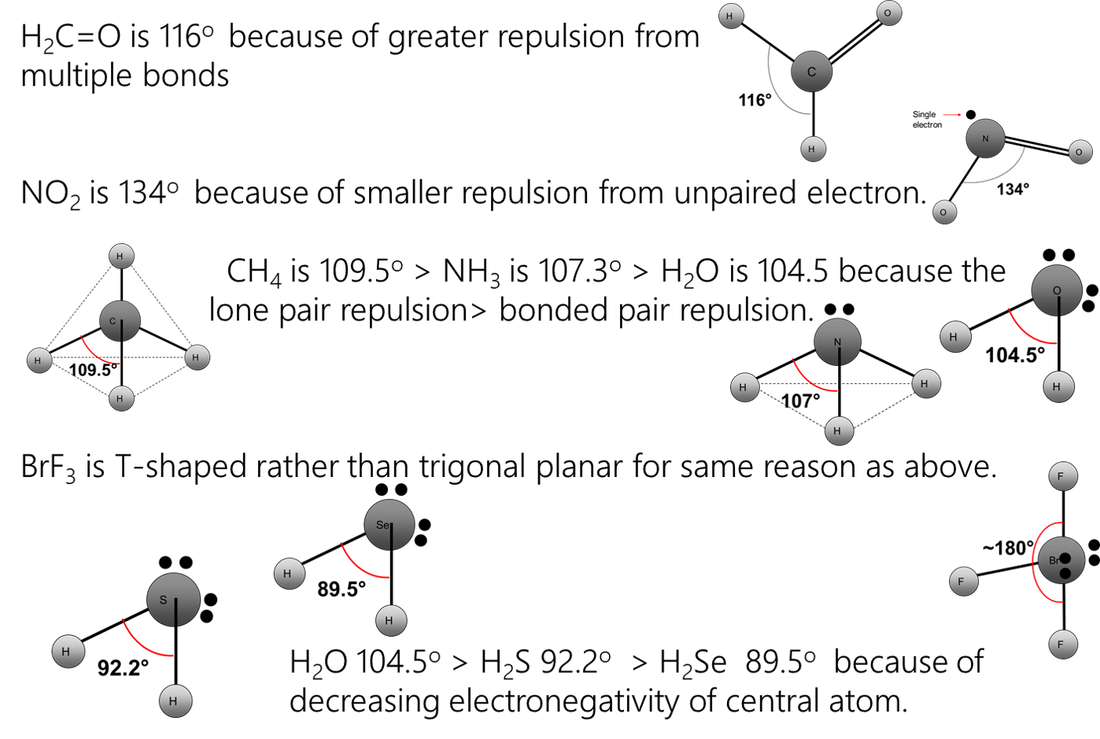
Repelling power of charge regions: triple bond > double bond > lone pair > single bond
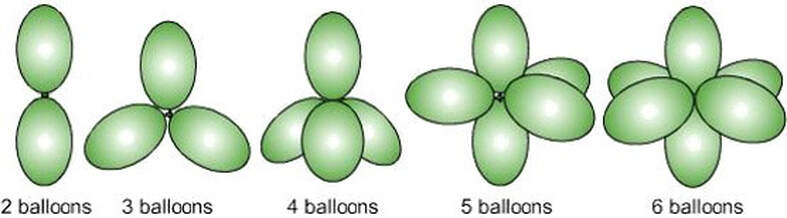
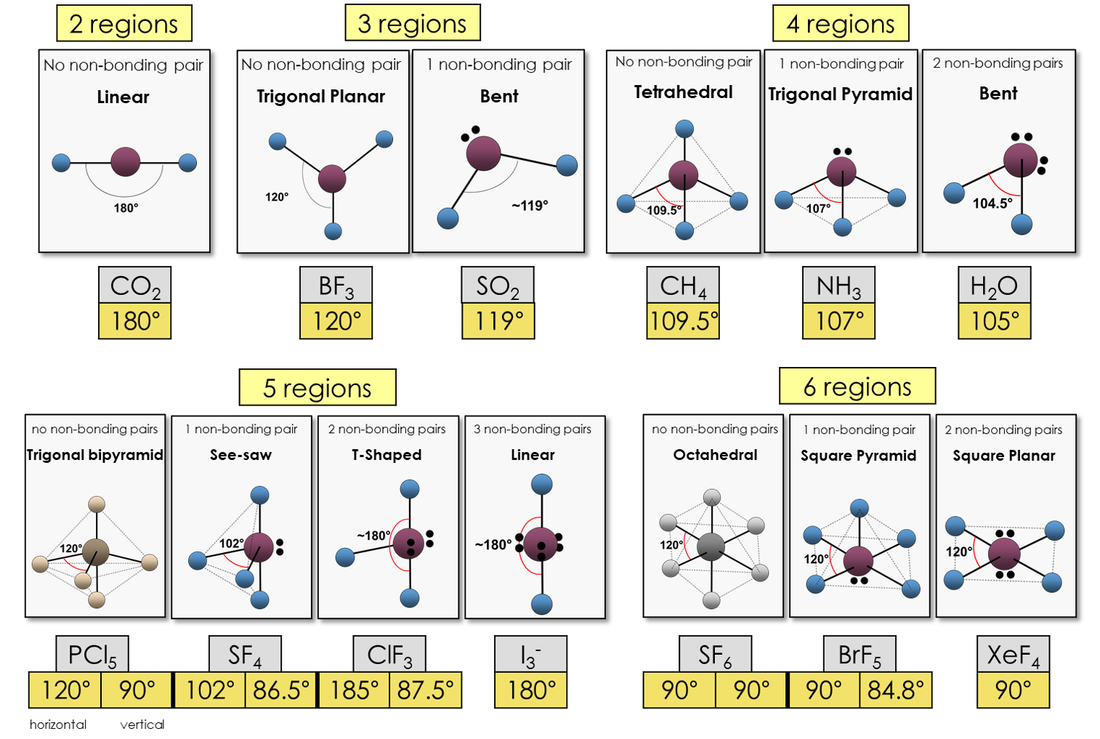
The balloons below represent the 'shape' of a molecule depending on how many regions form around a central atom.
Repelling power of charge regions: triple bond > double bond > lone pair > single bond
The balloons below represent the 'shape' of a molecule depending on how many regions form around a central atom.
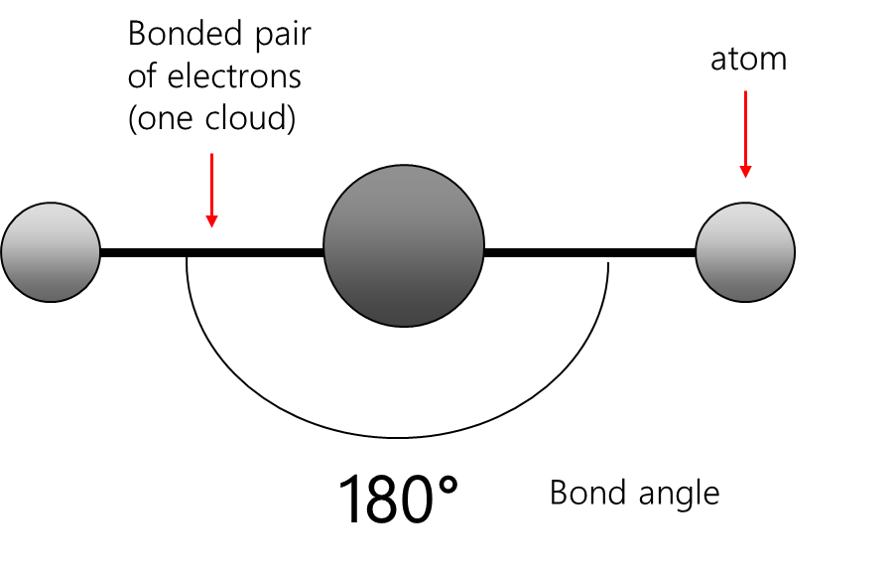
Linear
|
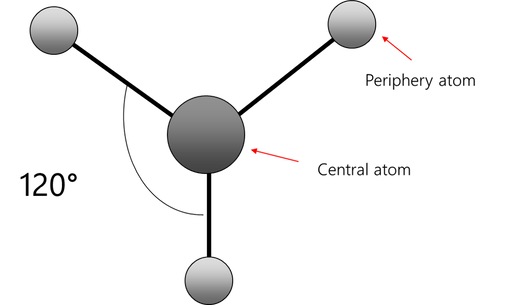
Trigonal Planar
|
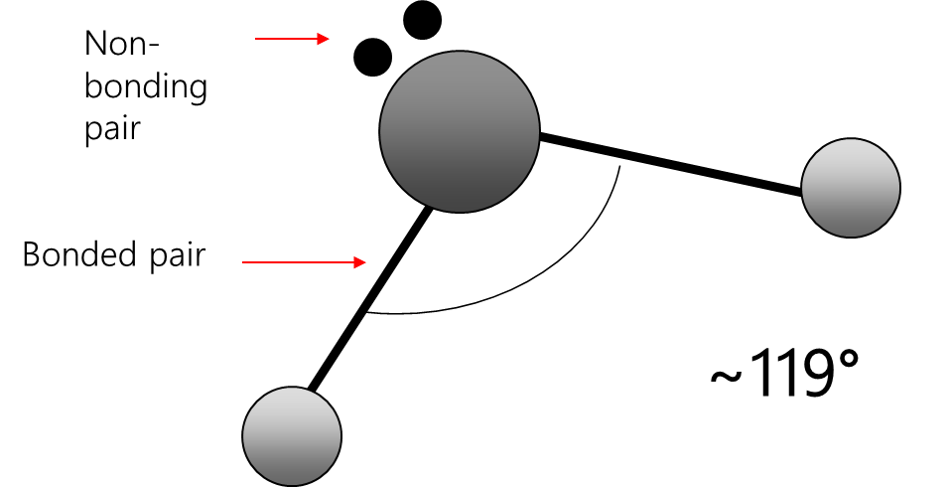
Bent
|
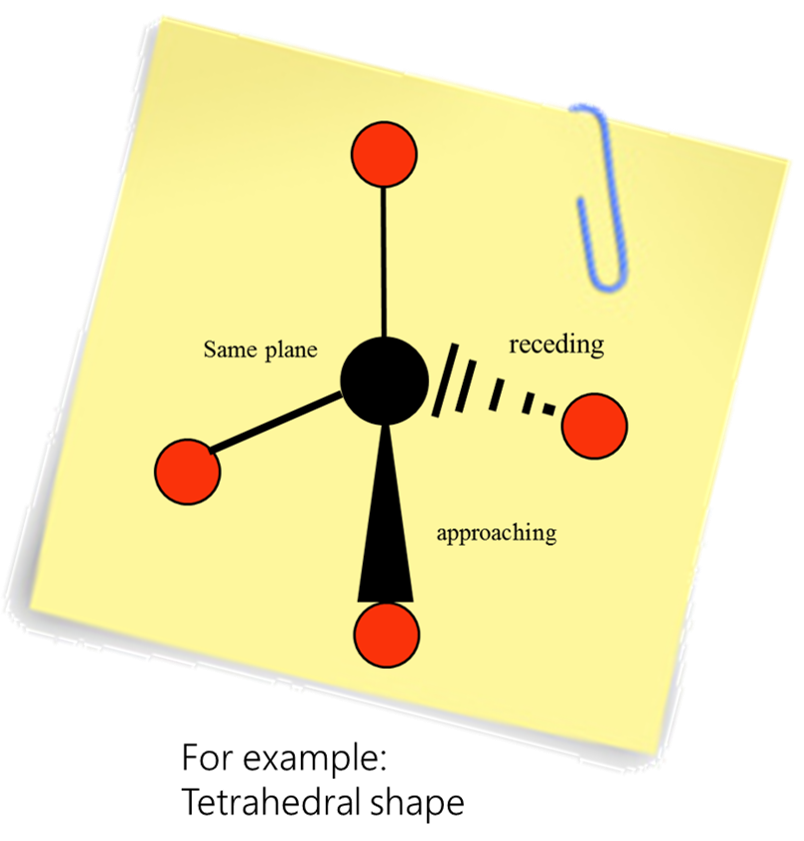
Tetrahedral
|
trigonal pyramid
|
bent
|
trigonal bipyramid
|
See-saw
|
t-shaped
|
linear
|
octahedral
|
Square Pyramid
|
square planar
|