Covalent Bonding
|
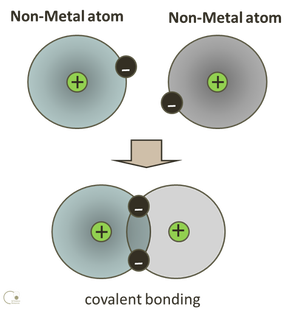
Covalent Bonding occurs when electrons are shared between neighbouring atoms. No ions are formed and there is no transfer of electrons. The compound formed is neutral with no charge. When the electronegativity is similar between atoms then the electrons will be shared evenly creating a non-polar molecule. If there is a significant electronegativity difference then dipoles will be created when the electrons orbit for a greater time around the more electronegative atom. This will create a polar molecule.
|
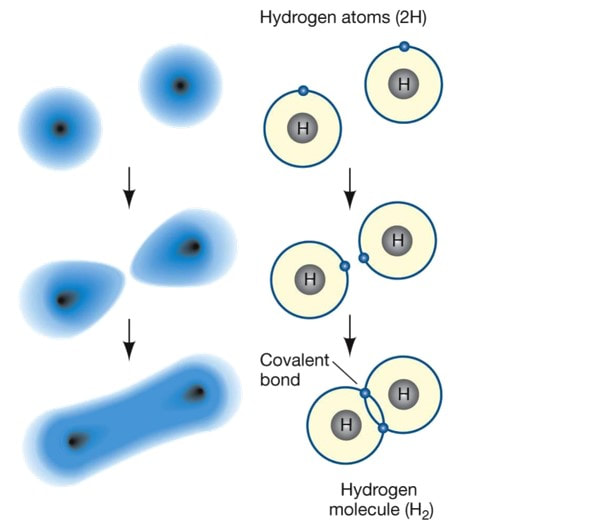
The valance electrons are involved in bonding. These electrons orbit in pairs. The negative charge of the electron pair will attract the positively charged nuclei of other atoms, and this holds the atoms together in a molecule.
|
The electron-pair must lie between the nuclei for the attraction to outweigh the repulsion of the two nuclei. This ‘sharing’ of electrons between atoms creates a covalent bond – giving both atoms the stability of a full outer shell.
Covalent bonds are normally formed between pairs of non-metallic atoms. Some covalent bonds involve only one pair of electrons and are known as single bonds. Other covalent bonds can involve two pairs of electrons; double bonds and three pairs of electrons; triple bonds. |
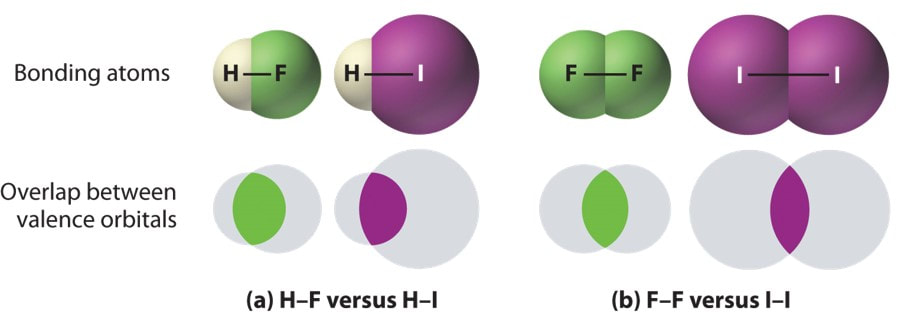
All covalent bonds are strong. That is it requires a large amount of energy to ‘break’ the bond. However, some covalent bonds are stronger than others. The greater the overlap of valence orbitals (the area the valence electrons orbit the nucleus) the stronger the bond.
Polarity and dipoles
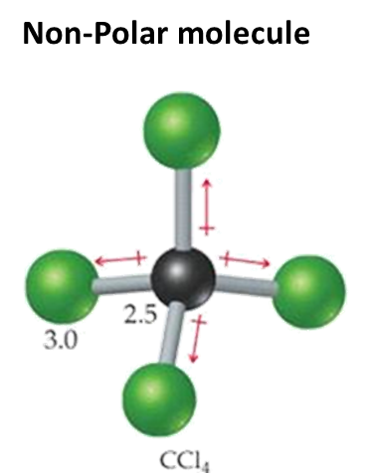
The bonded atoms have exactly the same amount of attraction to the shared electrons in the bonded pair. This is because their electronegativity is the same. This becomes a non-polar molecule with non-polar bonds. Example - Iodine molecule I2
Polar
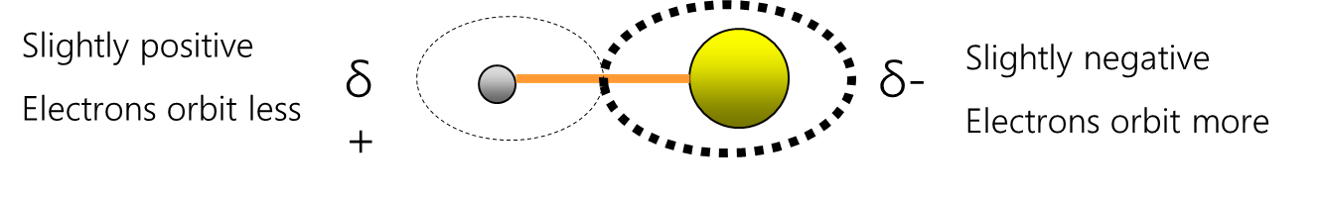
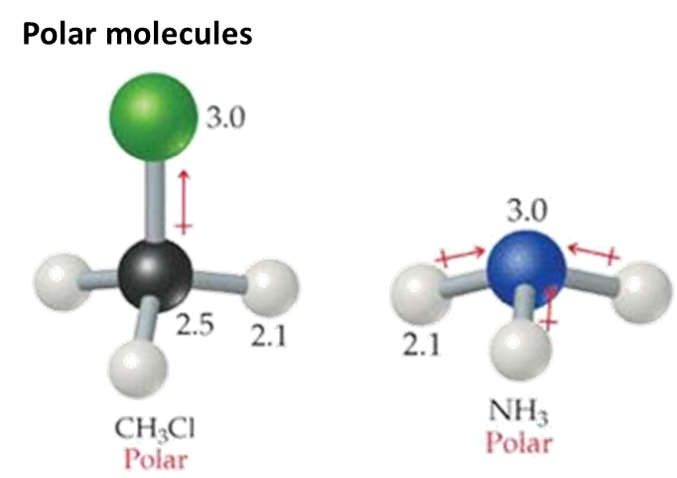
If two different atoms are bonded they form a polar bond, as there is an electronegativity difference and the valence electrons spend more time around the atom with the higher electronegativity value (that atom becomes slightly negative ) The atom that the valence electrons spend less time around becomes slightly positive.
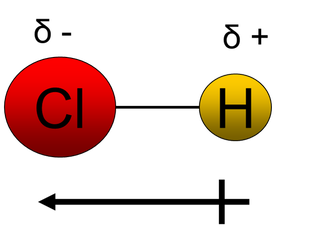
The atoms exert different levels of attraction for the orbiting electrons. That is because they may have different numbers of electron shells and different numbers of protons in their nucleus. This will cause an electronegativity difference and a dipole will form. These bonds become polar bonds. Example – hydrochloric acid HCl
If two different atoms are bonded they form a polar bond, as there is an electronegativity difference and the valence electrons spend more time around the atom with the higher electronegativity value (that atom becomes slightly negative ) The atom that the valence electrons spend less time around becomes slightly positive.
The atoms exert different levels of attraction for the orbiting electrons. That is because they may have different numbers of electron shells and different numbers of protons in their nucleus. This will cause an electronegativity difference and a dipole will form. These bonds become polar bonds. Example – hydrochloric acid HCl
Symmetry and Polarity
|
The overall polarity of a molecule with polar bonds depends upon whether the molecule is symmetrical or not and if all bonds are the same.
A symmetrical molecule (one where the centres of peripheral atoms coincide) becomes a non-polar molecule (only if all bonds are the same) – as the bond dipoles cancel out. |
|
An unsymmetrical molecule (one where the centre of peripheral atoms do not coincide) is a polar molecule, – as the bond dipoles do not cancel out.
If the bonds (central to peripheral) are different, then even if molecule is symmetrical the dipoles will not cancel out and therefore the molecule is polar overall |
Showing Polarity on a molecule
Solubility
The solubility of a substance is the amount of that substance that will dissolve in a given amount of solvent. Solubility is a quantitative term and varies depending on the solvent and the solute. The terms soluble and insoluble are relative. Some substances can be sparingly soluble where only the minutest percentage dissolves.
For a solute to dissolve the attraction to the solvent molecules must be stronger than the bonds holding the atoms/molecules of the solute together.
For a solute to dissolve the attraction to the solvent molecules must be stronger than the bonds holding the atoms/molecules of the solute together.
|
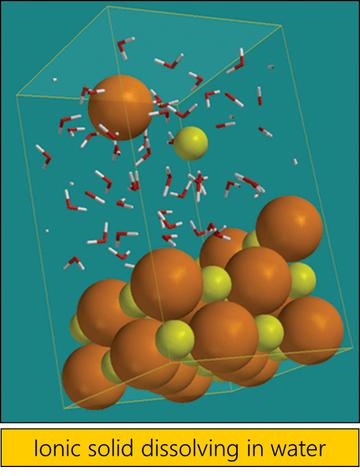
Polar substances dissolve polar substances.
e.g. Water, being polar attracts the molecules of other polar substances (e.g. HCl) and will dissolve them. Polar substances will not dissolve non-polar substances. e.g. Water, (polar) has a stronger attraction to itself than to non-polar molecules (e.g. cyclohexane) and will not dissolve them. Non-polar substances dissolve non-polar substances. e.g. Non polar solvents (like cyclohexane) attract non-polar solutes (like napthalene) by the same weak Van der Waals forces they attract themselves by and so will dissolve non-polar solutes. |