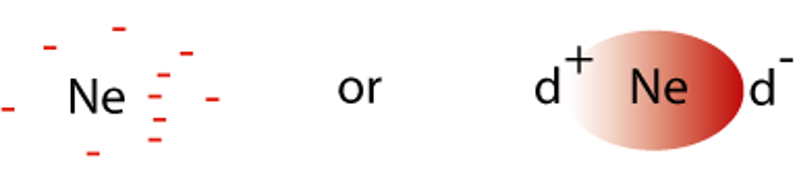Weak Inter-molecular Force
|
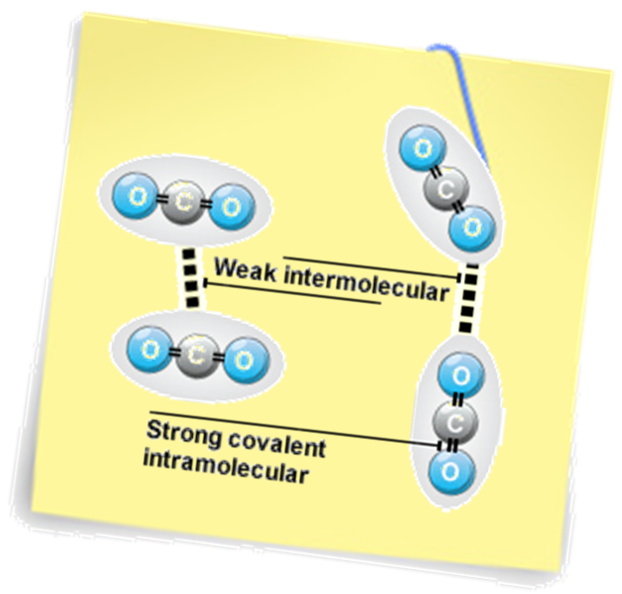
Weak intermolecular forces of attraction occur between molecular solids
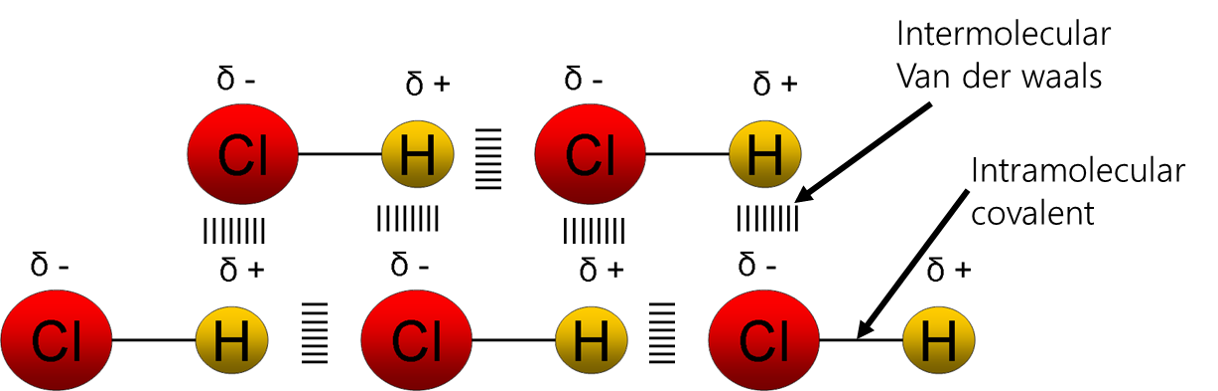
3 types of forces: Instantaneous dipole (ID-ID) – temporary dipole – all molecules have this type Permanent dipole (PD-PD) – permanent dipole – only polar molecules have this type in addition to ID-ID Hydrogen bonding (HB) – permanent dipole – molecules with H bonded to O, N or F have this type of bonding Note the distinction: Intra-molecular Forces: the strong bonding forces within a molecule. i.e. the covalent bonds holding the molecule together. Inter-molecular Forces: the weak bonding forces between molecules due to the attractions between partial charges. |
Instantaneous dipole (ID – ID) Attractions
The inert gases (group 18) are monatomic, and must be non-polar, yet the atoms attract together to form liquids and freeze to form solids. Likewise, non-polar molecules like cyclohexane condense and freeze. This suggests that some kind of bonding force operates between non-polar monatomic elements or non-polar molecules.
In any monatomic element or molecule, the electrons are moving rapidly. At any one instant, the arrangement of electrons about the nucleus will not be symmetric. This creates an instantaneous dipole (ID).
In any monatomic element or molecule, the electrons are moving rapidly. At any one instant, the arrangement of electrons about the nucleus will not be symmetric. This creates an instantaneous dipole (ID).
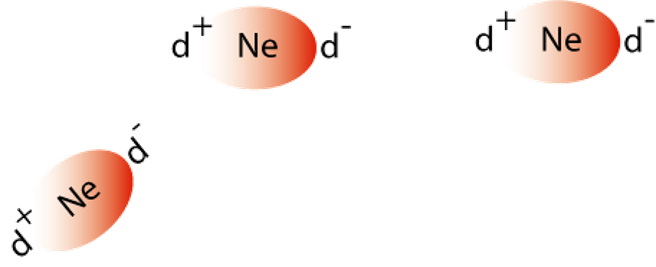
An atom with an ID will attract electrons in a neighbouring atom. This causes an induced dipole in this atom. The instantaneous dipole atom then attracts the induced dipole atom.
Permanent Dipole
|
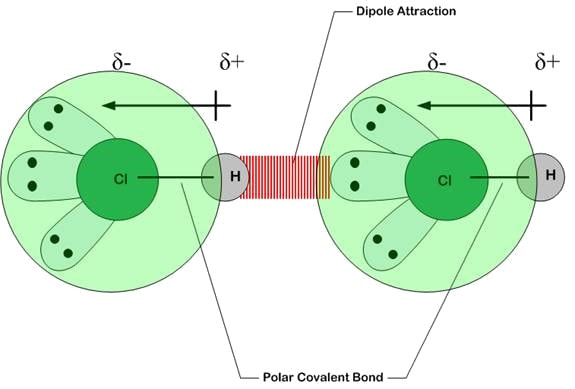
Permanent dipole (PD – PD) Attractions
Polar molecules have an imbalance of charge caused by an electronegativity difference between its constituent atoms. The weak molecular force always involves instantaneous dipole but in small molecules permanent dipole will play a larger part in the bonding. The slightly negative end of one molecule will be attracted to the slightly positive end of another molecule. The higher strength of the PD-PD bonding means that polar molecules will have a higher boiling/melting point to non-polar molecules (with only ID-ID bonding) of similar molar mass. |
The weakness of PD – PD bonding is revealed by the low melting points of PD molecular solids, by comparison with other types of solids.
e.g. hydrogen chloride (PD – PD) -114 °C acetone (PD – PD) -95 °C NaCl (ionic) 801 °C copper (metallic) 1085 °C
e.g. hydrogen chloride (PD – PD) -114 °C acetone (PD – PD) -95 °C NaCl (ionic) 801 °C copper (metallic) 1085 °C
Molecules that have side chains do not “pack” together so well as single chain molecules so cannot get as close to other molecules – all things being equal straight chained versions of molecules tend to have higher MP and BP