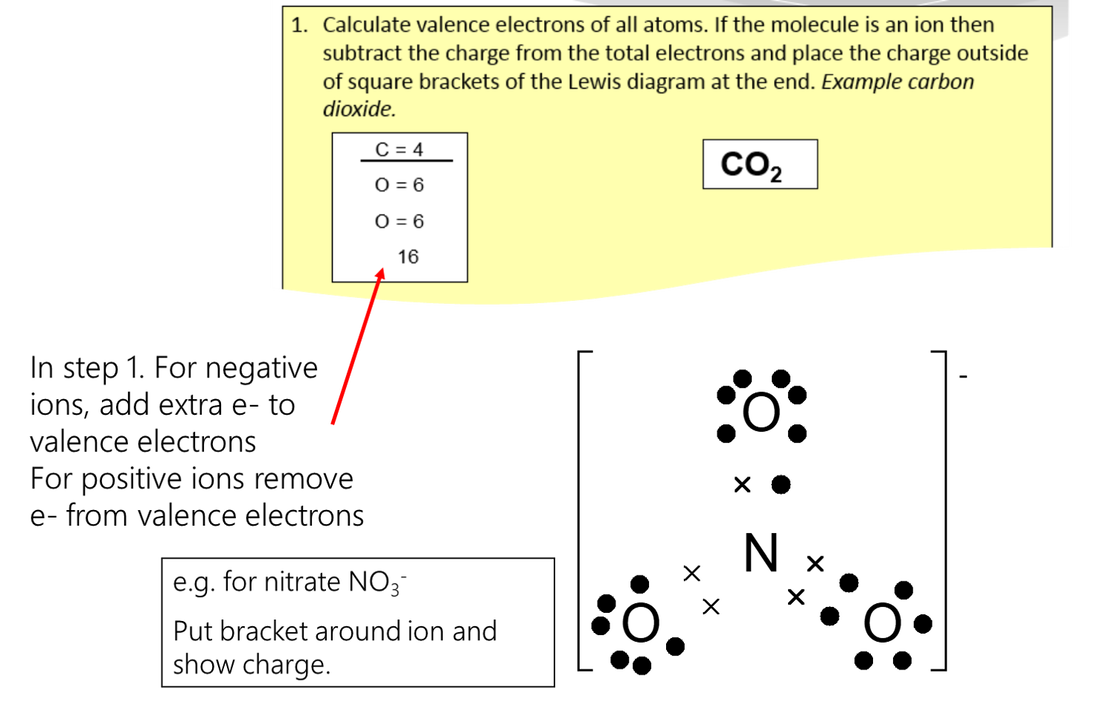
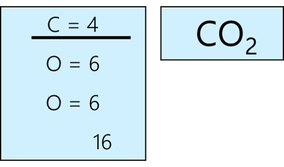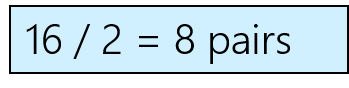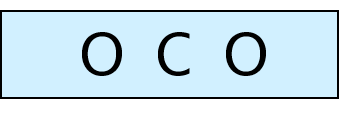Lewis Diagrams – Steps to drawing
7. If there are any remaining pairs place them around the outside of the central atom.
8. Rearrange non-bonded pairs into bonded pairs if the central atom does not have 4 pairs around it. i.e. create double, or triple bonds.
8. Rearrange non-bonded pairs into bonded pairs if the central atom does not have 4 pairs around it. i.e. create double, or triple bonds.
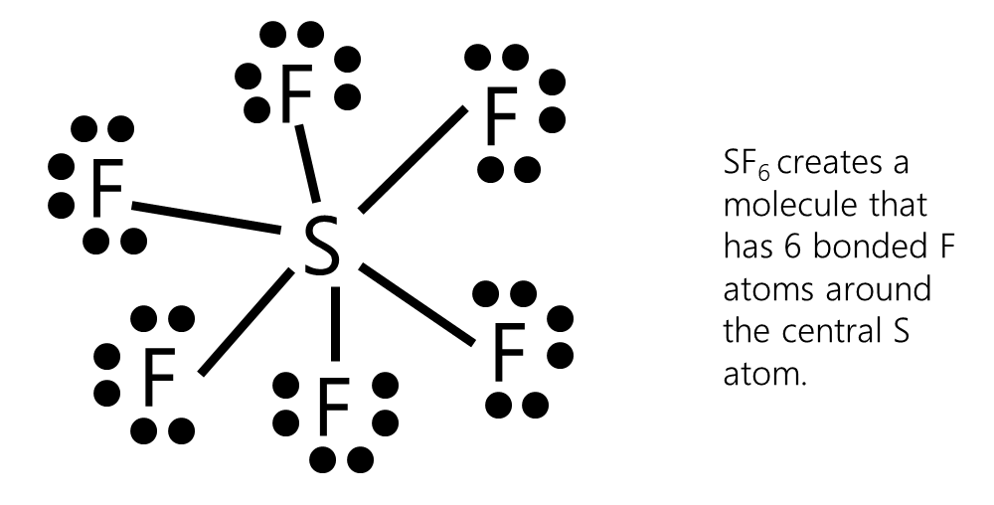
Expanded Octet
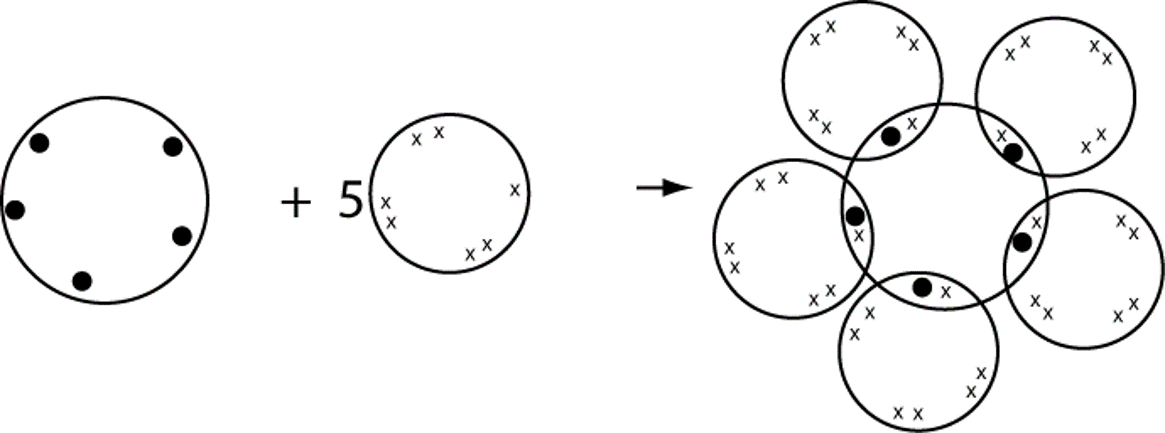
Third row elements (e.g., Al, Si, P, S, Cl) often have more than four valence shell orbitals filled with non-bonded Pairs and/or Bond Pairs; this is called "expanded Octet". Obviously, elements from the fourth and higher rows can also exhibit "expanded valence". EXAMPLE Phosphorus, electron configuration 2,8,5 can form up to 5 covalent bonds.
e.g. phosphorus pentachloride
P + 5Cl > PCl5
e.g. phosphorus pentachloride
P + 5Cl > PCl5
|
Rule of orbitals – exceptions to the rule
If there are extra non-bonded Pairs of electrons left after all of the periphery atoms are filled in accordance with the octet rule then they are placed around the central atom(s) according to the Rule of Orbitals. [extension] The Rule of Orbitals: the total number of non-bonded pairs and bonded pairs (NBP+BP) associated with an atom cannot exceed the number of Valence Shell Orbitals (VSO = n2 (n squared), where n is the row of the Periodic Table in which that atom resides). n = 1 (H): maximum VSE pairs (NBP+BP) = VSO = 1; n = 2 (B, C, N, O, F): maximum VSE pairs (NBP+BP) = VSO = 4 ("octet rule") n = 3 ((Al, Si, P, S, Cl): maximum VSE pairs (NBP+BP) = VSO = 9 etc. |
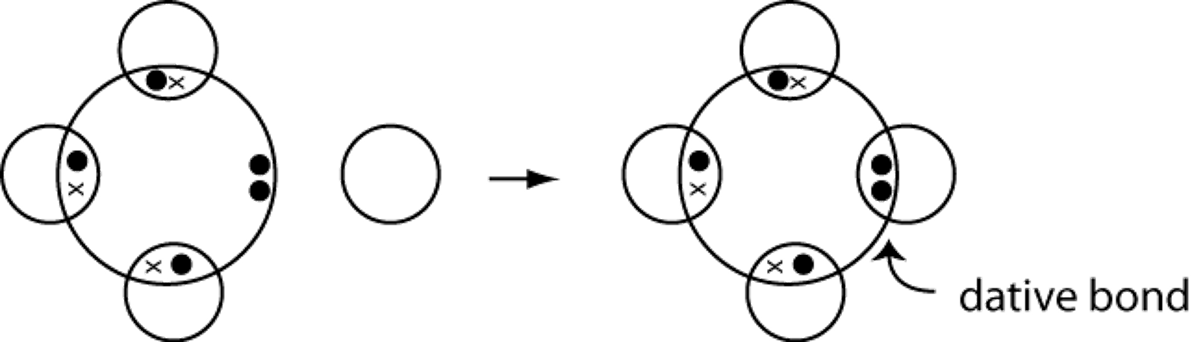
Dative Bonds
A dative bond occurs when one atom donates both electrons to form a single covalent bond.
e.g. an ammonium ion is made from an ammonia molecule plus a hydrogen ion.
NH3 + H+ > NH4+
e.g. an ammonium ion is made from an ammonia molecule plus a hydrogen ion.
NH3 + H+ > NH4+