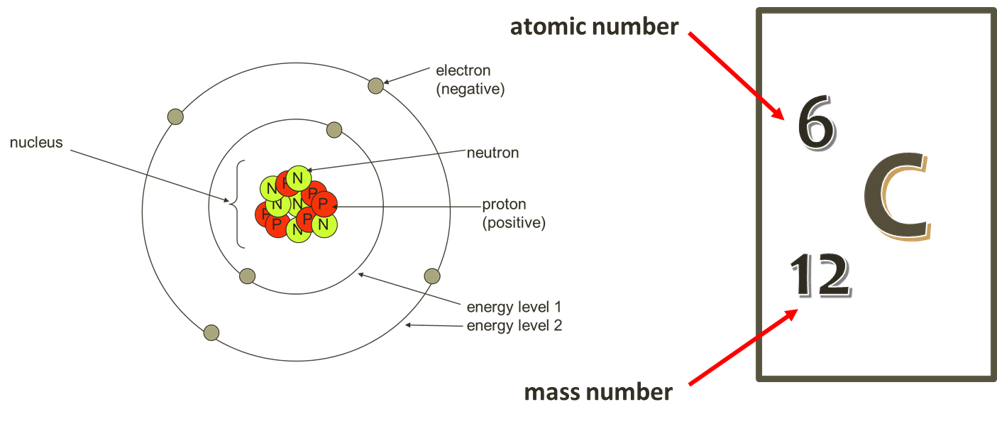
Atomic and Mass number
The atomic number is unique for each element, representing the number of protons in an atom’s nucleus.
In a uncharged atom, this also equals the number of electrons. The periodic table is arranged in order of an elements atomic number.
The mass number represents the average number of protons and neutrons in an atoms nucleus, of all possible isotopes with each particle equating to 1 mass unit (1amu).
In a uncharged atom, this also equals the number of electrons. The periodic table is arranged in order of an elements atomic number.
The mass number represents the average number of protons and neutrons in an atoms nucleus, of all possible isotopes with each particle equating to 1 mass unit (1amu).
Electrons orbit a nucleus of an atom at fixed energy levels.
|
An electron can be located in a fixed energy level around a nucleus of an atom.
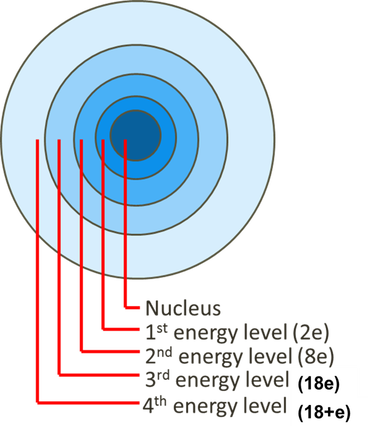
A ball bouncing down a flight of stairs provides an analogy for energy levels of electrons because a ball can only rest on each step, not between steps. Electrons move or ‘orbit’ around the nucleus in energy levels or shells. The energy levels further away from the nucleus are able to fit more electrons. The first energy level is filled first, followed by the second and so on until all the electrons (equal in number to the protons in an atom) are placed.
An electron can move up an energy level(s) if energy is absorbed, such as light or heat. When the electron moves back down to its lowest possible level the energy is released again, often in the form of light. |
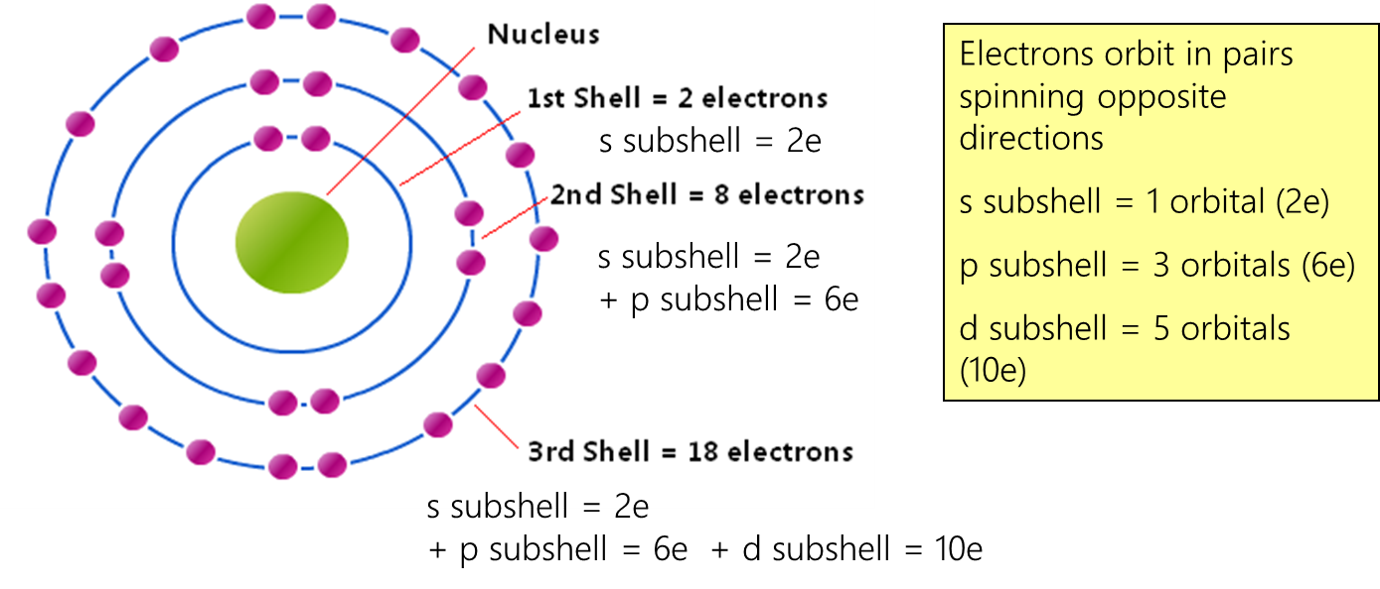
Electron orbitals
The major energy levels (shell) split into sub shells. Each sub shell level made up of one or more atomic orbitals. An orbital is an area of space with high probability of finding a particular electron pair. Electrons orbit in pairs spinning opposite directions.
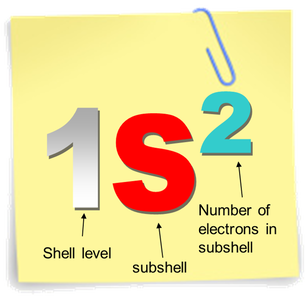
Electron Configuration notation
|
Even though the subshells are filled according to energy level, the notation is often written out in numerical order. 1s2,2s2,2p6,3s2,3p6,3d10,4s2,4p6…..
The sub shells are numbered according to the major shell/energy level they are in. Atoms and ions with the same electron configuration are said to be isoelectronic. |
As it is the valence shell that is of the most interest, the inner shells up to that point may be shown by the symbol for the previous inert gas element (neon) e.g. Cl [Ne] 3s2 3p5 rather than 1s2 2s2 2p6 3s2 3p5
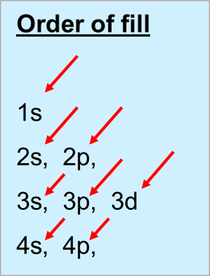
Order of fill
The arrangement of the electrons around the nucleus is known as its configuration. The Aufbau principle states that electrons always fill the shells with the lowest energy level first.
|
1. All of the orbitals hold up to 2 electrons which make up a pair (s1,p3,d5).
2. The electron pairs spin in opposite directions indicated with arrows 3. Electrons fill up orbitals with lower energy levels first 4. The most stable (lower energy) arrangement is for the sub levels to be filled with a single electron first (with the same direction spin) before pairing electrons. (Hund’s rule) Place electrons singly first showing upwards arrow. |